APGOT-OV1 (VIP)
GO TO LINK
Vinorelbine in Relapsed Platinum Resistant or Refractory C5 High Grade Serous, Endometrioid, or Undifferentiated Primary Peritoneum, Fallopian Tube or Ovarian Cancer
Pre Screening
Treatmenu phase
Follow-up
Current Recruitment Status
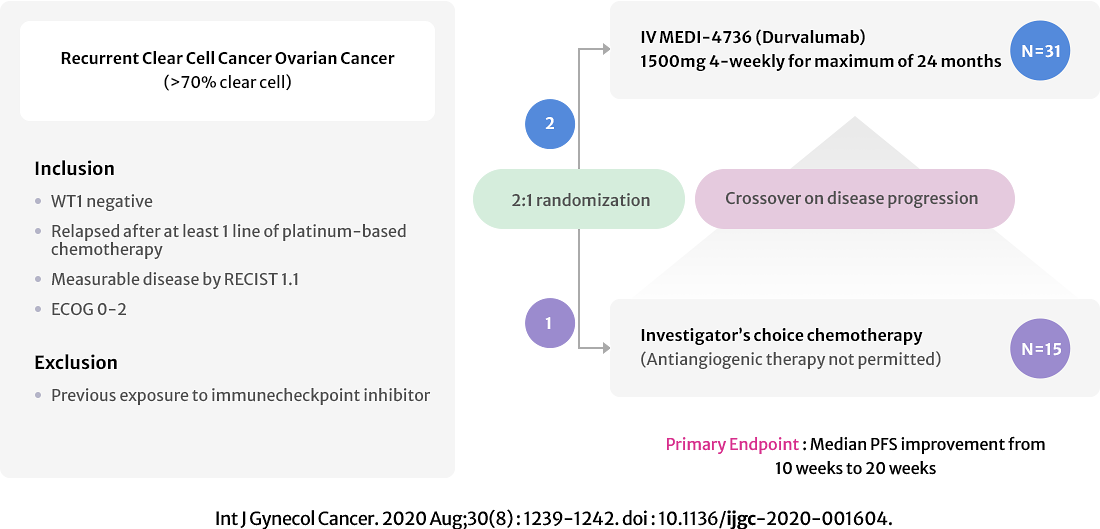
APGOT-OV2 (MOCCA)
GO TO LINK
A Multicentre Phase II Trial of Durvalumab Versus Physician's Choice Chemotherapy in Recurrent Ovarian Clear Cell Adenocarcinomas
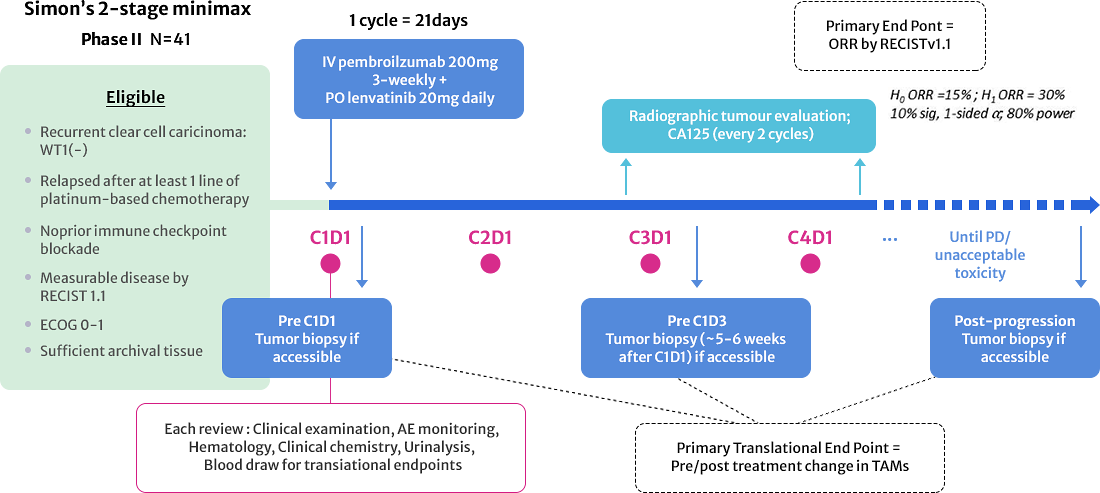
APGOT-OV3 (LARA)
GO TO LINK
Trial of Lenvatinib Plus PembrolizumAb in Recurrent Gynecological Clear Cell Adenocarcinomas (LARA)
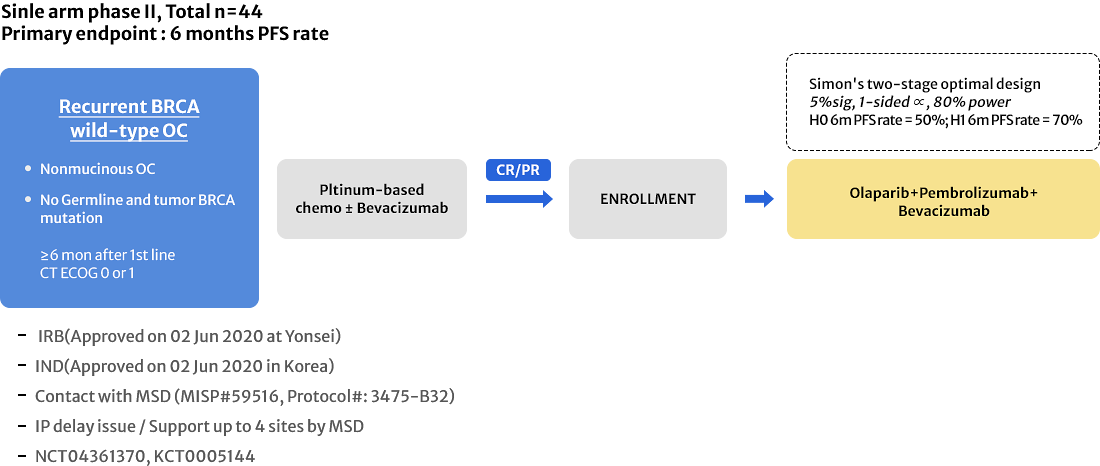
APGOT-OV4 (OPEB-01)
GO TO LINK
Olaparib Maintenance With Pembrolizumab & Bevacizumab in BRCA Non-mutated Patients With Platinum-sensitive Recurrent Ovarian Cancer
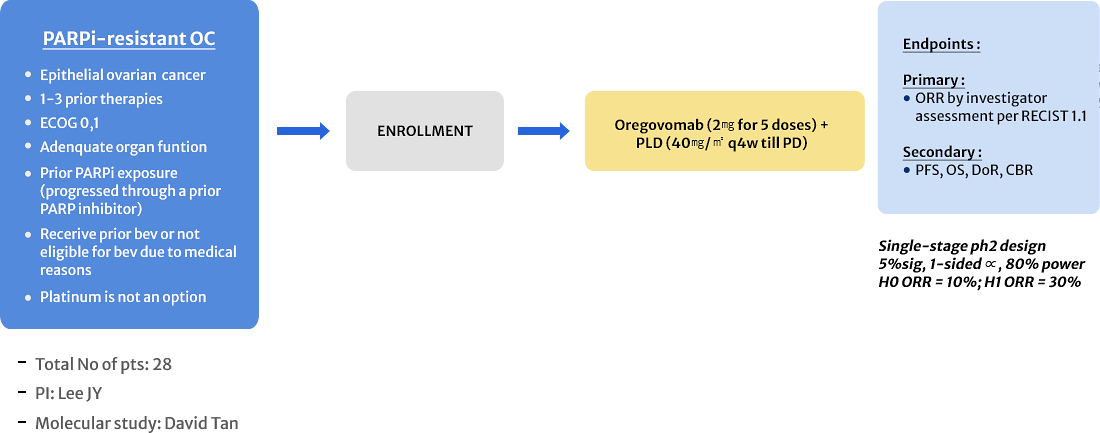
APGOT-OV6 (OPERA)
GO TO LINK
Oregovomab and PLD in PARP Inhibitor Resistant Ovarian, Fallopian Tube, or Primary Peritoneal Cancer Patients Not Candidate for Platinum Retreatment
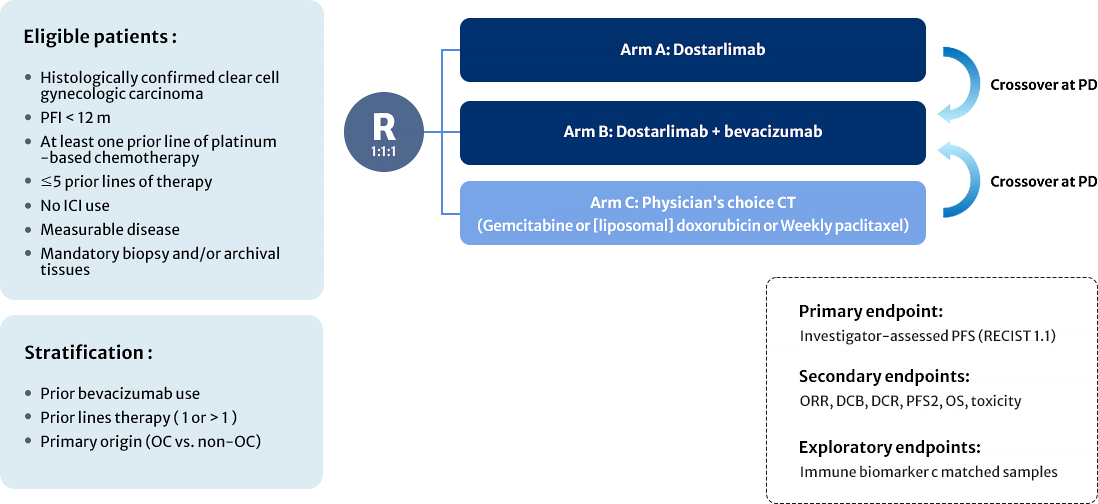
APGOT-OV7 (DOVE)
GO TO LINK
A Three-arm Randomized Phase II Study of Dostarlimab Alone or With Bevacizumab Versus Nonplatinum Chemotherapy in Recurrent Gynecological Clear Cell Carcinoma: DOVE (APGOT-ov7/ ENGOT-ov80 Study) (DOVE)
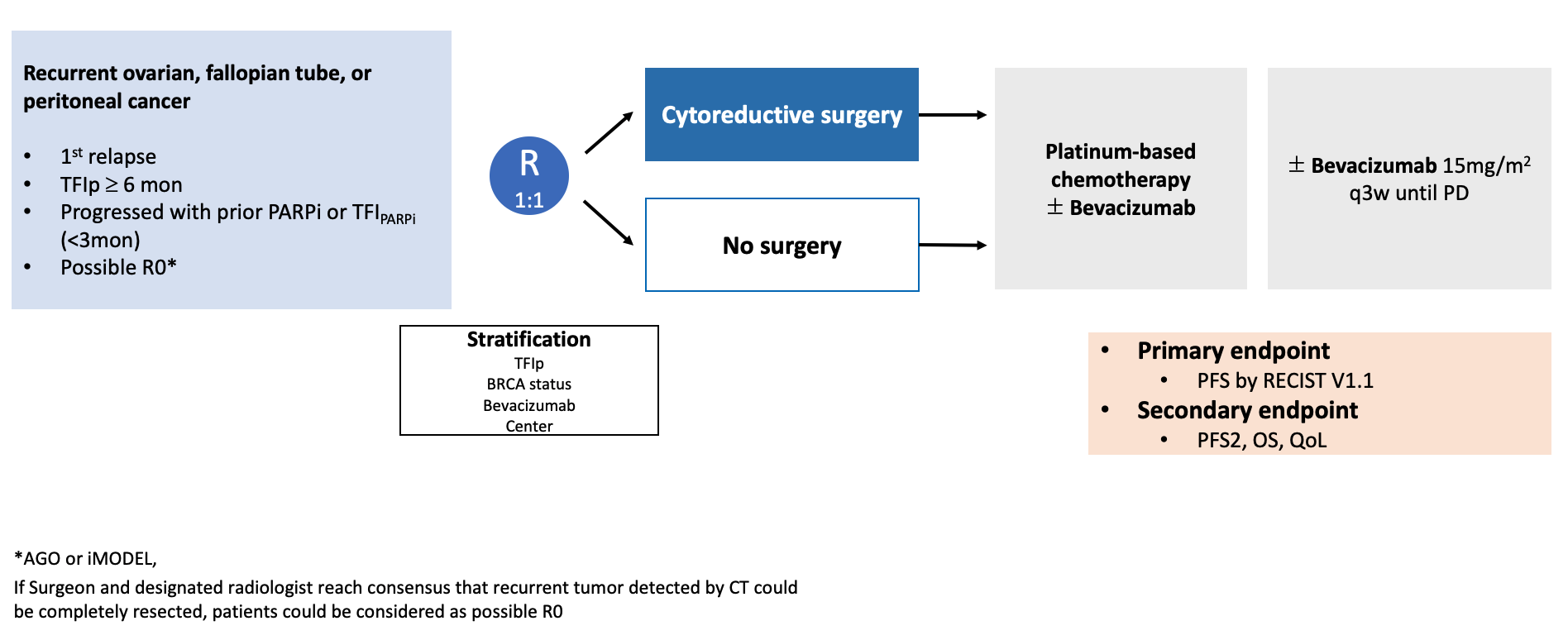
APGOT-OV11 (SOCCER-P)
GO TO LINK
Secondary Cytoreductive Surgery in Patients With Relapsed Ovarian Cancer Who Have Progressed on PARP Inhibitor Maintenance (SOCCER-P)